Absence of nuclei and transcellular cytoskeletons, human erythrocytes rely on the cytoskeleton system to maintain their unique biconcave shape. With extreme deformation and stability, erythrocytes can travel through the circulation system millions of times during their 120-day lifespan without damage. In addition, the erythrocyte membrane often exhibits a tank tread-like motion when erythrocytes were subject to shear stress. Especially, with the stop of shear flow, a shape memory phenomenon exists, which means the specific regions of a deformed erythrocyte, e.g., the dimple region, were always formed by the same parts after it returned to its original shape. Although the biconcave shape and shape memory characteristics of human erythrocytes are closely associated to the organization of cytoskeleton, the underlying molecular mechanism is still a mystery.
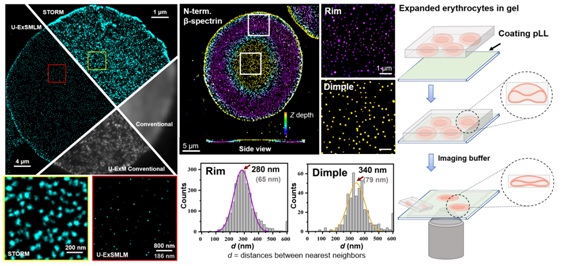
Expansion microscopy (ExM) has been established to be compatible with existing super-resolution microscopy to further improve the resolution, including SIM (Ex-SIM) and STED (Ex-STED). However, attempts to combine single-molecule localization microscopy (SMLM) with ExM remain rare and have not been optimized, mainly due to the sharp shrinkage of hydrogel upon the application of imaging buffer. Recently, the research group of Prof. Pan Leiting and Prof. Xu Jingjun from Nankai University developed an ultrastructure expansion single-molecule localization microscopy (U-ExSMLM) based on poly-l-lysine adhering to avoid gel shrinkage. The effective resolution of U-ExSMLM reached the molecular level of ~ 6 nm. The ultrastructure of the cytoskeleton in human erythrocytes preserving the native biconcave morphology was investigated by U-ExSMLM. It was found that the the dimple region of erythrocytes had a longer nearest distance and lower density of spectrin skeletons than the rim region, which provided direct molecular resolution evidence for the asymmetry of the cytoskeleton in biconcave human erythrocytes.
The fascinating biconcave morphology of erythrocytes has attracted a lot of theoretical and experimental researchers to elucidate its formation mechanism since its discovery, yet the final conclusion remains unreached. Late Pro. J. Hoffman, academician of the American Academy of Sciences, centrifuged the hypotonic-induced spherical erythrocyte ghosts onto the slides. After adjusting the osmotic pressure, he observed that when recovering biconcave shapes, most of the ghosts were lying flat on the coverslip, rather than upright or randomly distributed. Therefore, there was probably a dense band in equatorial plane of the sphered erythrocyte ghosts, leading to oriented attachment to the coverslip during centrifugation. He suggested that this macro-level evidence of cytoskeleton asymmetry is responsible for biconcave shape of human erythrocytes, and pointed out that super-resolution imaging is expected to prove this asymmetry at the micro-level (PNAS, 2016, 113, 14847-14851; PNAS, 2018, 115, 1641-1645). In the present work, U-ExSMLM visually revealed the molecule-level asymmetry of spectrin cytoskeleton in biconcave human erythrocytes, verifying Hoffman's hypothesis, as well as proposed a promising tool for the study of membrane-proximal cellular components.
To sum up, the present work proposed a strategy for the combination of SMLM and ExM relied on poly-l-lysine adhering. The developed method of U-ExSMLM reached the molecular resolution of ~6 nm, and directly revealed the asymmetry of the erythrocyte cytoskeleton, providing a molecular explanation for understanding the unique biconcave morphology and shape memory phenomenon in human erythrocytes.
The Paper Link: https://onlinelibrary.wiley.com/doi/full/10.1002/smtd.202201243



